Multiple Choice
Identify the
letter of the choice that best completes the statement or answers the question.
|
|
|
1.
|
The number of atoms in a mole of any pure substance is called
a. | its atomic number. | c. | its mass number. | b. | Avogadro’s number. | d. | its gram-atomic
number. |
|
|
|
2.
|
What can be said about 1 mol Ag and 1 mol Au?
a. | They are equal in mass. | b. | They contain the same number of
atoms. | c. | Their molar masses are equal. | d. | They have the same atomic
mass. |
|
|
|
3.
|
An Avogadro’s number of any element is equivalent to
a. | the atomic number of that element. | c. | 6.022 ´ 1023 particles. | b. | the mass number of that
element. | d. | 12 g of that
element. |
|
|
|
4.
|
A formula that shows the simplest whole-number ratio of the atoms in a compound
is the
a. | molecular formula. | c. | structural formula. | b. | ideal formula. | d. | empirical
formula. |
|
|
|
5.
|
To determine the molar mass of an element, one must know the
element’s
a. | Avogadro number. | c. | number of isotopes. | b. | atomic number. | d. | average atomic
mass. |
|
|
|
6.
|
What is the empirical formula for a compound that is 31.9% potassium,
28.9% chlorine, and 39.2% oxygen?
a. | KClO2 | c. | K2Cl2O3 | b. | KClO3 | d. | K2Cl2O5 |
|
|
|
7.
|
A compound contains 64 g of O and 4 g of H. What is the empirical formula for
this compound?
|
|
|
8.
|
A molecular compound has the empirical formula XY3. Which of the
following is a possible molecular formula?
|
|
|
9.
|
A compound’s empirical formula is NO2. If the molecular mass is
92 amu, what is the molecular formula?
|
|
|
10.
|
What is the sum of the atomic masses of all the atoms in a formula for a
compound?
a. | ionic mass | c. | atomic mass | b. | molar mass | d. | actual mass |
|
|
|
11.
|
The molar mass of LiF is 25.94 g/mol. How many moles of LiF are present in 10.37
g?
a. | 0.3998 mol | c. | 2.500 mol | b. | 1.333 mol | d. | 36.32 mol |
|
|
|
12.
|
What is the percentage composition of CuCl2?
a. | 33% Cu, 66% Cl | c. | 65.50% Cu, 34.50% Cl | b. | 50% Cu, 50% Cl | d. | 47.27% Cu, 52.73%
Cl |
|
|
|
13.
|
The molar mass of CS2 is 76.15 g/mol. How many grams of
CS2 are present in 10.00 mol?
a. | 0.13 g | c. | 10.00 g | b. | 7.614 g | d. | 761.5 g |
|
|
|
14.
|
The molar mass of NH3 is 17.03 g/mol. How many moles of
NH3 are present in 107.1 g?
a. | 0.1623 mol | c. | 6.289 mol | b. | 3.614 mol | d. | 107.1 mol |
|
|
|
15.
|
Which of the following compounds have the same empirical formula?
a. | NO2 and N2O5 | c. | C5H10 and
C3H6 | b. | SO3 and
NO3 | d. | H2O and H2O2 |
|
|
|
|
|
|
16.
|
A farmer makes up a new counting unit called a
“cluckster.”
If the farmer had 3 clucksters of chicken eggs and 3 clucksters of quail
eggs, what could you say about the ratio of their masses?
a. | The clucksters of chicken eggs will be 16 times heavier than the 3 clucksters of
quail eggs | c. | The clucksters of quail eggs will be 16 times heavier than the 3 clucksters of
chicken eggs | b. | The clucksters of chicken eggs will be 3 times heavier than the 3 clucksters of quail
eggs | d. | The clucksters of
chicken eggs will be 438 times heavier than the 3 clucksters of quail
eggs |
|
|
|
Use the following
passage and graphs to answer the following three questions.
To practice
solving percentage composition problems, a student made a series of circle graphs showing the
composition of a sample of carbon dioxide (CO2). Graph A is based on a sample that is 100% CO2. Graph B represents the mass fractions of
a 100.0-g sample of CO2. Graph
C represents the mass fractions of a 23.5-g sample of CO2.
|
|
|
17.
|
Which graph displays an incorrect
composition relationship?
a. | Graph A | c. | Graph
C | b. | Graph B | d. | All of the graphs
are correct. |
|
|
|
18.
|
Based on the data shown in Graph C, how many atoms
of carbon are there in a 23.5-g sample of CO2?
a. | 2.26 X
1023 atoms | c. | 6.02 X
1023 atoms | b. | 3.21 X 1023 atoms | d. | 1.18 X
1024 atoms |
|
|
|
19.
|
What percentage (on a mass basis) of the sample in
Graph C is oxygen (O2)?
a. | 6.41% | c. | 27.28%
| b. | 23.49% | d. | 72.72% |
|
|
|
20.
|
Avogadro's number of representative particles is equal to one ____.
a. | kilogram | c. | kelvin | b. | gram | d. | mole |
|
|
|
21.
|
All of the following are equal to Avogadro's number EXCEPT ____.
a. | the number of atoms of bromine in 1 mol Br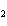 | b. | the number of atoms of gold in 1 mol
Au | c. | the number of molecules of nitrogen in 1 mol N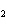 | d. | the number of molecules of carbon monoxide in 1
mol CO |
|
|
|
22.
|
How many moles of tungsten atoms are in 4.8 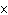 10 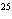 atoms of tungsten?
|
|
|
23.
|
How many atoms are in 0.075 mol of titanium?
|
|
|
24.
|
Which of the following is NOT a true about atomic mass?
a. | The atomic mass is 12 g for magnesium. | b. | The atomic mass is the mass of one mole of
atoms. | c. | The atomic mass is found by checking the periodic table. | d. | The atomic mass is
the number of grams of an element that is numerically equal to the mass in
amu. |
|
|
|
25.
|
What is the molar mass of (NH 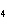 ) 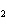 CO 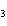 ? a. | 144 g | c. | 96 g | b. | 138 g | d. | 78 g |
|