•So what is the name
of FeCl2?
of FeCl2?
•Chloride always has
a 1- charge, and there
are two, so…
a 1- charge, and there
are two, so…
•The positive guy must
have a total
2+ charge, and…
have a total
2+ charge, and…
•There is only one cation so it get the whole 2+ charge all to itself, so…
•It must be iron(II), so…
•It is iron(II) chloride
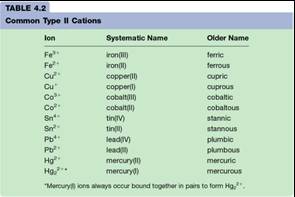
•Know Table 4.2 (the systematic is emphasized in this class!!)